How Electron Shells Shape Our World
Imagine a world where skyscrapers had no floors, where buildings stood on thin air. Sounds impossible, right? In the atomic world, electron shells play the role of these invisible floors, organizing electrons around the nucleus of an atom. These shells aren’t physical structures but rather energy levels where electrons reside, dictating how atoms interact, bond, and create everything around us—from the air we breathe to the smartphones we use.
Understanding electron shells is key to unlocking the mysteries of chemistry, physics, and even biology. But what exactly are they, and why do they matter? Let’s take a closer look at these tiny yet powerful structures that govern the behavior of matter.
The Story of Electron Shells: A Dance of Energy Levels
In 1913, Danish physicist Niels Bohr proposed a revolutionary idea—electrons do not just randomly orbit the nucleus of an atom. Instead, they occupy discrete energy levels, or electron shells. Think of these shells like the floors of a high-rise building, where electrons can move up or down but never hover in between.
Atoms naturally strive for stability, and their electron arrangements define their chemical properties. The outermost shell, called the valence shell, determines how atoms interact. If this shell is full, the atom is stable (like the noble gases—helium, neon, argon). If it’s not full, the atom becomes reactive, eagerly seeking to bond with other atoms to complete its shell.
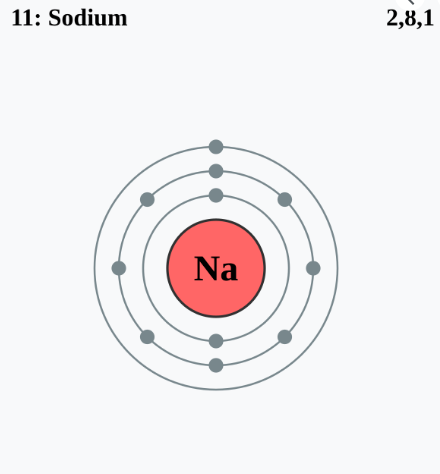
But how do these shells fill up? Scientists follow a simple rule: the first shell holds up to 2 electrons, the second up to 8, the third up to 18, and so on. This structured filling of shells explains the periodic table’s organization and why elements behave the way they do.
Why Electron Shells Matter: From Chemistry to Everyday Life
The impact of electron shells extends far beyond textbooks. Here’s how they influence the world around us:
1. Chemical Reactions and Bonding
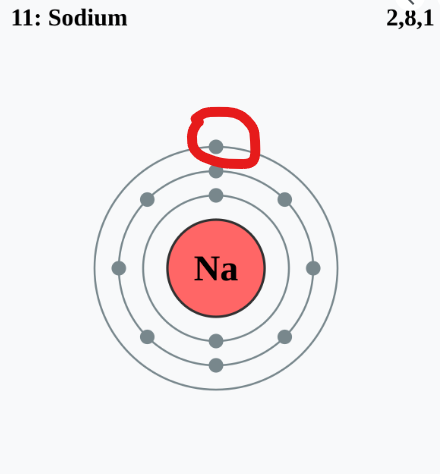
Ever wonder why salt (NaCl) forms so easily? Sodium (Na) has one lonely electron in its outer shell, while chlorine (Cl) needs one more to complete its shell. When sodium donates its electron to chlorine, they both achieve stability, forming a strong ionic bond. This principle of electron exchange governs how molecules form—giving us everything from water (H₂O) to complex proteins in our bodies.
2. Electricity and Conductivity
Metals like copper and gold conduct electricity so well because their outer electron shells are loosely held. These free-moving electrons create electrical currents, enabling the function of power grids, circuits, and electronic devices. Without this property, our modern world of technology wouldn’t exist.
3. Energy and Light Emission
When electrons jump between shells, they absorb or emit energy, often in the form of light. This phenomenon explains everything from the glow of neon signs to the twinkle of stars. In fluorescent bulbs, electrons are excited to higher shells and then drop back, releasing brilliant light in the process.
4. Medical Applications: X-Rays and Radiation
In medicine, electron shells play a vital role in imaging and treatment. X-rays work because high-energy electrons knock inner-shell electrons out of atoms, causing the emission of radiation that helps create detailed images of bones and tissues. Similarly, radiation therapy uses controlled electron disruptions to target and destroy cancer cells.
Making It Simple: An Everyday Analogy
Imagine a hotel with multiple floors. Guests (electrons) can only stay on specific floors (shells). The first floor (closest to the nucleus) fills up first before guests move to the next available floor. But there’s a twist—if the top floor isn’t full, guests feel restless, looking for other guests to form groups (chemical bonds). This is exactly how atoms behave, always striving for balance.
The Future: How Understanding Electron Shells Can Drive Innovation
Scientific breakthroughs in electron shell manipulation are already shaping the future. Quantum computing, nanotechnology, and even fusion energy research depend on the precise control of electron configurations. By understanding how electrons behave, scientists are designing more efficient batteries, cleaner energy sources, and next-generation materials that could revolutionize industries.
Conclusion: Why It Matters to You
Electron shells may be invisible to the naked eye, but they define the very essence of matter. From the food we eat to the technology we use, these tiny energy levels dictate how atoms interact and shape our world. The next time you turn on a light, use your phone, or take a deep breath, remember—it’s all possible because of the unseen but powerful organization of electron shells.
Curious to learn more about atomic structures and how they impact your daily life? Subscribe to our newsletter for more engaging science insights!



